GCI PowerTools for Electronic Signatures
GCI PowerTools for OpenText Electronic Signatures™ ensures process governance and eliminates the cost and effort by extending OpenText Electronic Signature to sign more than one document in the same workflow and enable control of signature pages and watermarks.

About the product
- Process and sign multiple documents within a single electronic signature workflow
- Dynamically identify which documents within a workflow to convert / watermark / sign
- Merge multiple source documents into a single signed document
- Add additional custom metadata to the signing page
- Simplify signing process for executives and other less frequent Content Suite users
Facilitate compliance with 21 CFR Part 11
OpenText Electronic Signatures enables compliance with 21 CFR Part 11 and PowerTools for Electronic Signatures extends the functionality, making it easier for organizations to comply.
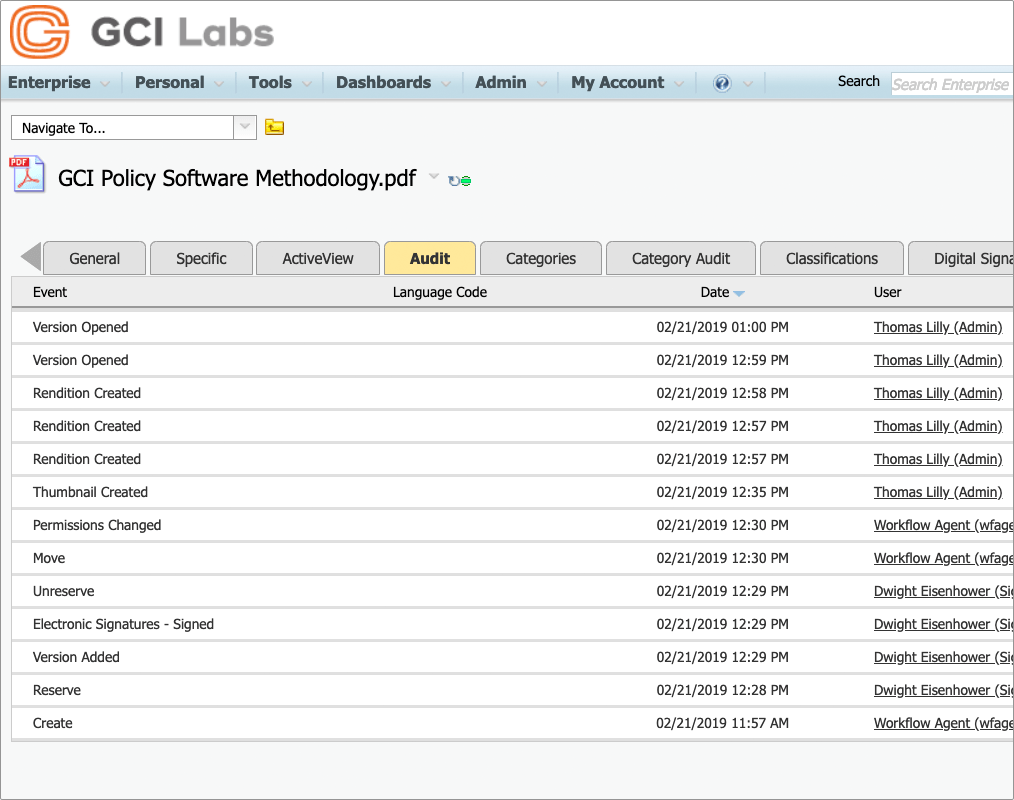
Electronic signatures for Content Suite
Approve and sign controlled documents directly within OpenText Content Suite and Extended ECM platforms. The whole process can be completed at minimum effort to end-users and a complete audit history is maintained.
Simultaneously sign multiple documents
Attach multiple documents to any individual electronic signing process, so that more than one document can be reviewed and signed simultaneously.
Assemble and merge documents ready for signing
Documents can be automatically assembled and merged into a final document which itself can then be approved and signed.
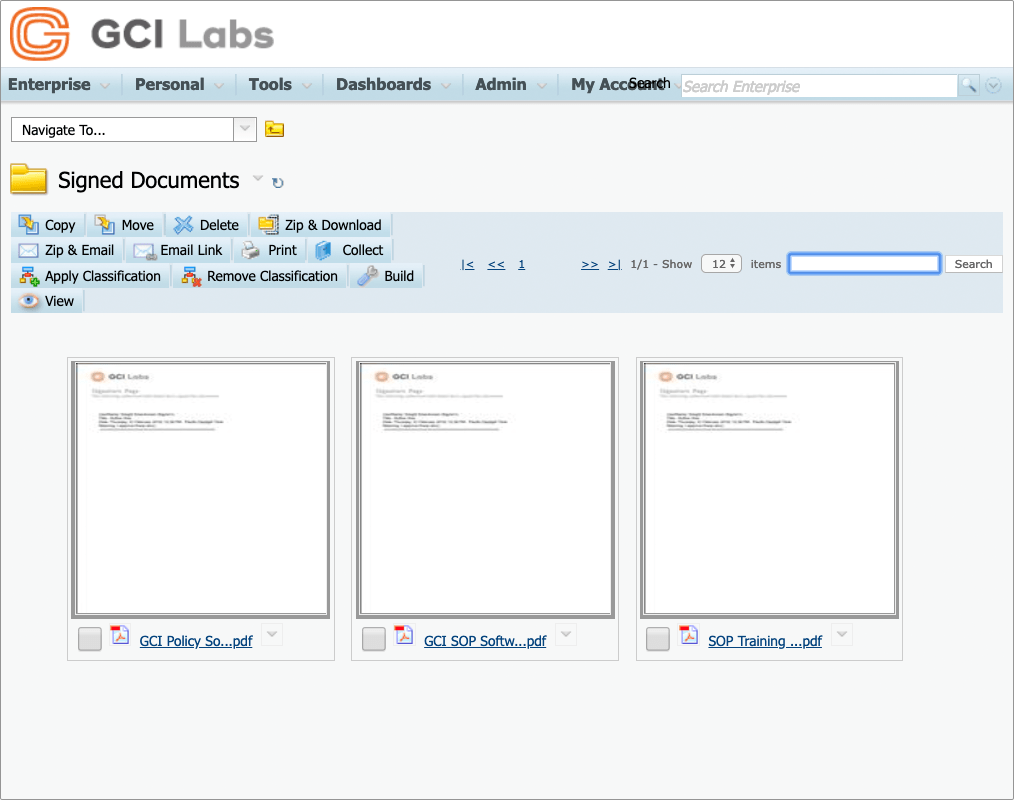
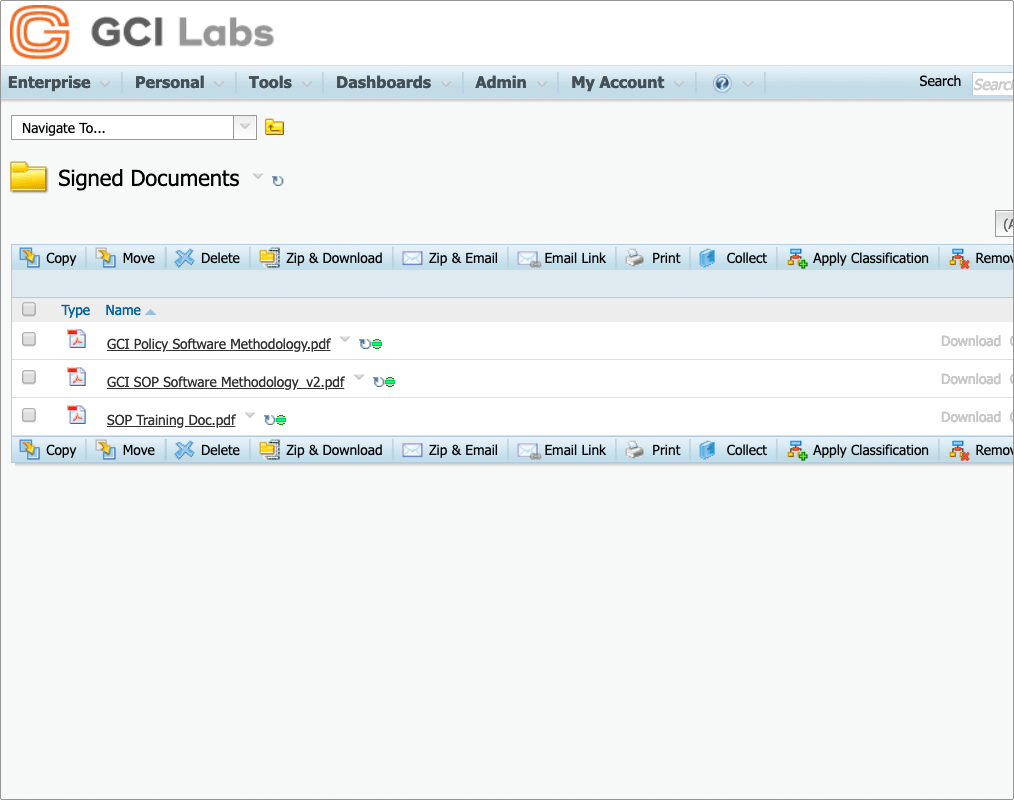
Convert signed documents to PDF
Any or all documents in a signing workflow can be converted to PDF, watermarked, and signed enabling your business process to be automated with minimum effort.
Securely archive signed documents
Once signing is complete, the original documents can either be left in their initial location or moved to an archive or other area. Likewise, signed documents can be left or moved as needed.
A simplified signing experience
Through optional integration with GCI PowerTools for Workflow, the entire workflow interface can be readily tailored and redesigned around each individual signing process.
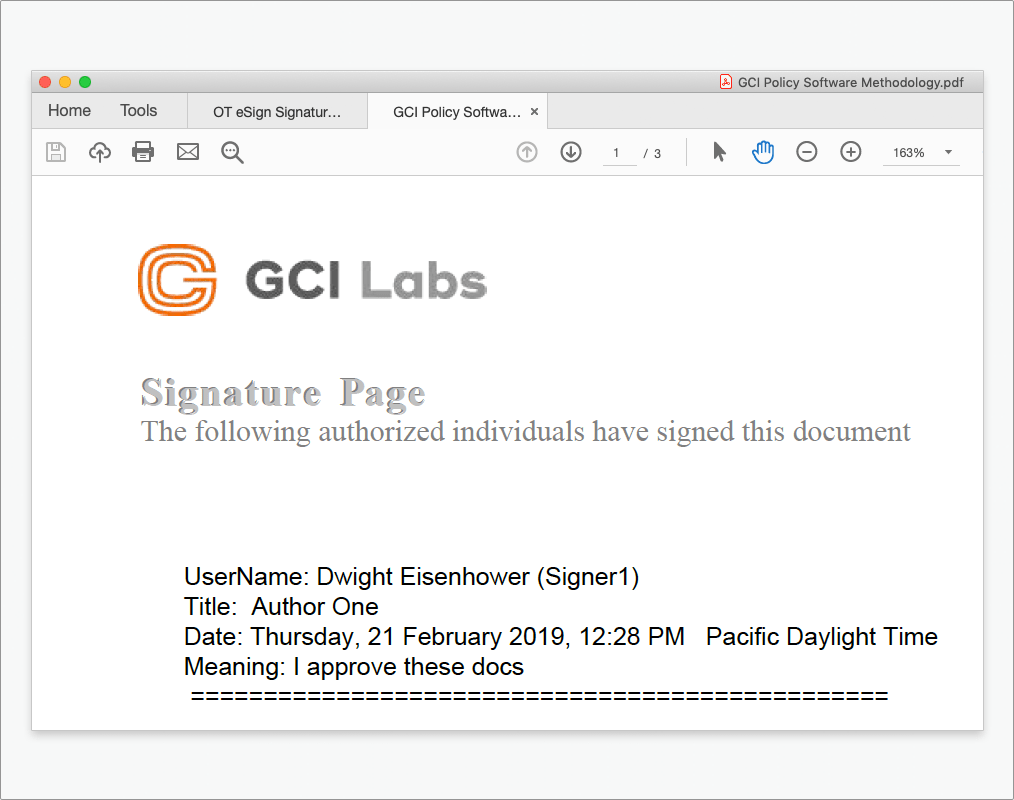
Powerful metadata stamps
Create customized signing pages containing organization-specific meta-tags to ensure all the information you need is captured in the format you want to see it.
Signing for regulated industries
PowerTools for Electronic Signatures is designed with the needs of regulated industries at the forefront. It is used to sign documents within life science, energy and financial services organizations and is deployed in certified environments.
Get further information
If you need to electronically sign documents for FDA or other regulatory compliance, then PowerTools for Electronic Signatures is the most comprehensive solution available. For further details or a personalized demonstration contact Global Cents.

